Ageing is related to a dynamic process of ongoing microvascular injury, such as blood-brain barrier disruption, impaired hemodynamics and clearance, i.e., removal of brain protein and metabolite waste products. These processes ultimately lead to synaptic and neural network dysfunction, but could be counterbalanced by continuous microvascular repair, which in turn should be related to maintenance of synaptic and network functioning. We propose, that balance between microvascular injury and repair as well as resistance against microvascular injury in response to physiological stimuli cumulatively fail with increasing age (denoted as “microvascular brain aging”), which feeds into reduced cognitive flexibility and function.
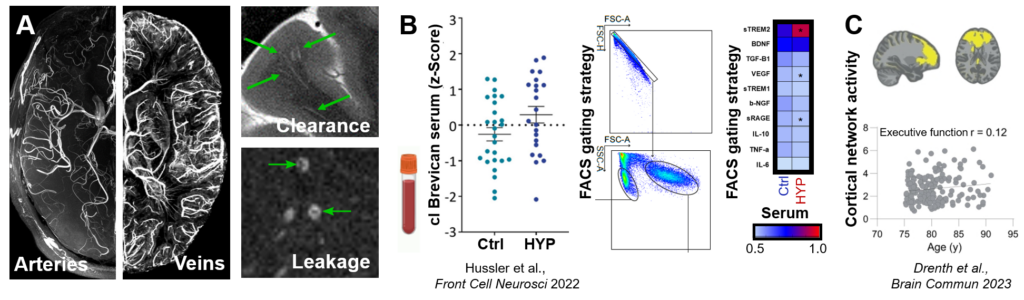
Figure 1. A. Ultra-high-resolution MRI of the small arteries (ToF MRA) and veins (QSM) (left) and of enlarged white matter/transcortical perivascular spaces (T2) as well as small vessel wall leakage in terms of BBB breakage (contrast enhanced T1) (right). B. Blood-based biomarker measures of ECM (cleaved (cl.) brevican, ELISA, left) and NVU markers or cytokines (FACS, right) in controls (Ctrl) and hypertension (HYP). C. Frontal network activity applying resting state functional MRI and its relationship to executive function in ageing.
Abbreviations: BBB, blood-brain barrier; cl, cleaved; Ctrl, controls; ECM, extracellular matrix; ELISA, enzyme-linked immunosorbent assay; FACS, fluorescence activated cell sorting; HYP, hypertension; MRA, magnetic resonance angiography; MRI, magnetic resonance imaging; NVU, neurovascular unit; QSM, quantitative susceptibility mapping; ToF, time of flight.

Project B2: S. Schreiber, Ph. Arndt, R. Previn, P. Müller, Ch. Piechowiak, S. Henneicke
Hence, in close interaction with project B1, we aim to study in rodents (B1) and humans (B2) cortical and hippocampal synaptic function and network connectivity as a function of “microvascular brain ageing”. We specifically focus on the question, how this relationship is influenced by age-related reduced microvascular resistance against physical stress or prolonged recovery. We further particularly investigate how age-related impaired microvascular resistance/recovery and associated declined synaptic and network function can be restored through targeted pharmacological treatment.
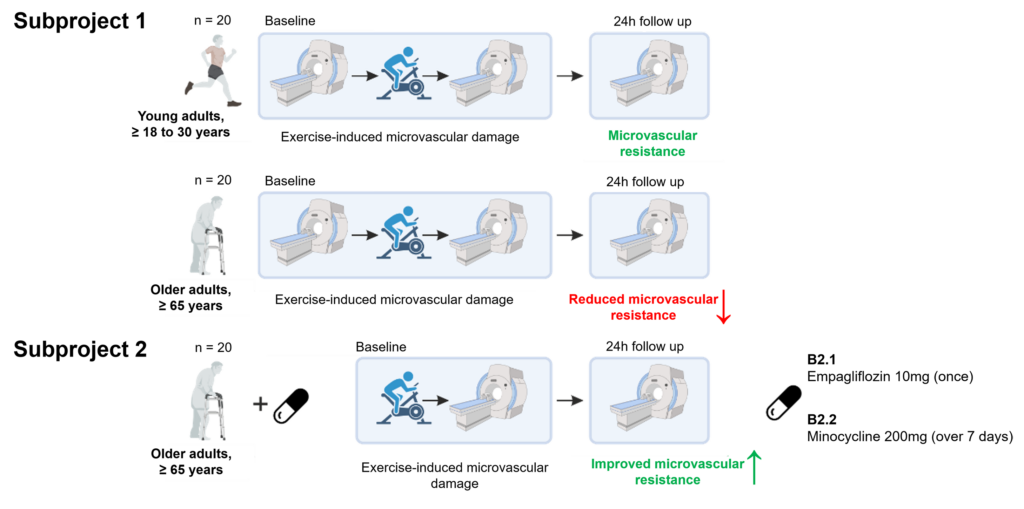
Figure 2. Study protocol in project B2.





Project B2: R. Previn, S. Henneicke, H. Mattern, P. Müller, S. Schreiber
Find out more about the working group by clicking here.
For more information on Hendrik Mattern’s research click here.
PhD thesis (Rahul Previn)
The human brain’s resting state is not as restful as it seems, as interconnected regions dynamically shift and reconfigure, impacting brain function, plasticity, and potential interventions. These complex dynamics are essential to brain function because they promote plasticity and provide prospective pathways for therapies aimed at better understanding and improving cognitive functions. To explore the effects of acute exercise on these dynamic resting-state networks, we conducted a study using functional magnetic resonance imaging (fMRI) on 20 healthy young subjects. The subjects underwent fMRI scans at three time points: pre-exercise (resting state), immediately post-exercise (resting state), and 24 hours after exercise (resting state). During the exercise session, participants engaged in a cycling exercise for a short duration to elevate blood flow, which can potentially disrupt the microvascular blood-brain barrier (BBB). Damage to the microvascular BBB can lead to the infiltration of harmful substances and cells into the brain, contributing to various neurological disorders. Previous studies in mice have shown that inducing blood flow can cause BBB damage (Doe et al., 2022), but it is challenging to replicate the same in healthy young humans. Therefore, we used cycling exercise as an extensive exercise that can disturb blood flow and potentially damage the BBB. To achieve reliable results, we employed the CONN toolbox for meticulous preprocessing of the MRI data and also collected physiological data, including pulse rate and heart rate, from the subjects. By integrating these preprocessed MRI and physiological data within the CONN toolbox, we aimed to explore major resting-state networks using advanced analysis techniques. Our goal is to understand how acute exercise impacts the dynamic networks of the resting brain, which can provide valuable insights for cognitive function and therapeutic applications, especially in the context of healthy aging and brain differences. In the future, we plan to screen older subjects to analyze the effect of extensive exercise on microvascular health.
ORCID ID: https://orcid.org/0000-0002-1853-7346
PUBLON ID: https://publons.com/researcher/1661965/rahul-previn
